Safety of sodium oxybate
Adverse Effects
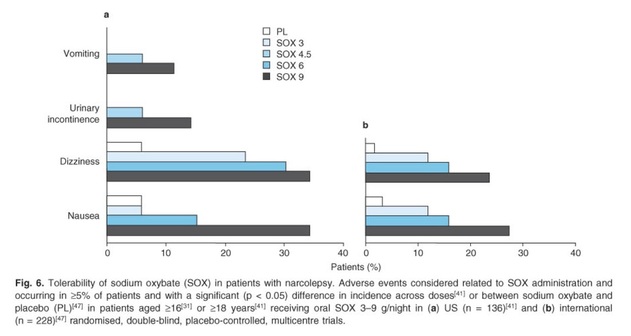
The following are the most common adverse events that were observed in trials that evaluated sodium oxybate in patients with narcolepsy that were conducted in the US and internationally:
o Vomiting
o Urinary incontinence
o Dizziness
o Nausea
o Bed-wetting
o Sore throat
These adverse events were considered to be related to sodium oxybate administration and occurred in >5% of patients in trials. There was a significant (p<0.05) difference in incidence across doses between sodium oxybate and placebo. Various trials noted that the frequency of adverse events appears to be dose-related. More severe adverse effects such as impaired consciousness and respiratory depression can but rarely occur, even at therapeutic doses; the risk is increased with supra-therapeutic doses.
Studies have also shown that there does not seem to be any rebound cataplexy upon abrupt discontinuation of treatment, when patients had stopped taking sodium oxybate at the end of the trial; thus, the risk of “status cataplecticus” often observed upon cessation of TCAs and SSRIs do not seem to be an issue with sodium oxybate when taken under approved and appropriate prescribing conditions.
Unexpected death during sleep has been described in at least three cases associated with use of sodium oxybate. One death appears associated with sodium oxybate, with extremely high blood levels of GHB due to intentional overdose. Although postmortem GHB levels in two other deaths were within therapeutic range, a direct cause and effect from this event cannot be established. The authors explained that the incidences may have been attributed to the respiratory depression caused by sodium oxybate alone or in combination, including simultaneous use of sedative hypnotics, obstructive sleep apnea and obesity.
Abuse potential
In the 1990s, GHB gained a reputation as a ‘club drug’ because of its use at raves and dance parties, and as a date rape drug because of its reports of intoxication and amnesia. These reports, as well as other reports of abuse and misuse of sodium, led to a retrospective review of postmarking adverse event reports related to sodium oxybate from 15 countries (including the US, EU, and Canada) from 2002 to 2008 concluded the following:
o The illicit use of GHB in frequent, repeated, supratherapeutic doses (18 – 250 g/day) has been associated with dependence; however, in patients receiving therapeutic doses of sodium oxybate (i.e. 4.5 g – 9 g/night), there is no clear evidence that dependence exists, although the possibility cannot be excluded
o When used according to prescribing and monitoring guidelines, sodium oxybate has been shown to be a safe and effective treatment for cataplexy and EDS in narcolepsy; it is associated with a very low incidence of misuse, abuse, and dependence
o U.S. data indicate that the prevalence of illicit GHB use is very low and has declined since 2000; it is thought to be much lower than that of the major drugs of abuse and club drugs such as ecstasy.
Special populations/precautions
The following are important considerations to keep in mind with respect to the use of sodium oxbate in special populations:
Contraindications
Concurrent use of Xyrem (sodium oxybate) with sedative hypnotic agents or alcohol is contraindicated. Xyrem is contraindicated in patients with succinic semialdehyde dehydrogenase deficiency. This rare disorder is an inborn error of metabolism variably characterized by mental retardation, hypotonia, and ataxia, and causes GHB to accumulate in the blood.
In the 1990s, GHB gained a reputation as a ‘club drug’ because of its use at raves and dance parties, and as a date rape drug because of its reports of intoxication and amnesia. These reports, as well as other reports of abuse and misuse of sodium, led to a retrospective review of postmarking adverse event reports related to sodium oxybate from 15 countries (including the US, EU, and Canada) from 2002 to 2008 concluded the following:
o The illicit use of GHB in frequent, repeated, supratherapeutic doses (18 – 250 g/day) has been associated with dependence; however, in patients receiving therapeutic doses of sodium oxybate (i.e. 4.5 g – 9 g/night), there is no clear evidence that dependence exists, although the possibility cannot be excluded
o When used according to prescribing and monitoring guidelines, sodium oxybate has been shown to be a safe and effective treatment for cataplexy and EDS in narcolepsy; it is associated with a very low incidence of misuse, abuse, and dependence
o U.S. data indicate that the prevalence of illicit GHB use is very low and has declined since 2000; it is thought to be much lower than that of the major drugs of abuse and club drugs such as ecstasy.
Special populations/precautions
The following are important considerations to keep in mind with respect to the use of sodium oxbate in special populations:
- In patients with liver dysfunction, the starting dose of sodium oxybate should be half of the usual starting dose, and adverse events should be closely monitored as the dose is titrated.
- Xyrem (sodium oxybate) should only be ingested at bedtime.
- For at least 6 hours after ingesting sodium oxybate, patients must not engage in hazardous occupations or activities requiring complete mental alertness or motor coordination, such as operating machinery, driving a motor vehicle, or flying an airplane.
- When patients first start taking Xyrem or any other sleep medicine, they should use extreme caution while performing tasks that could be dangerous or requires full mental alertness; they should continue to do so until they know whether the medicine will still have some carryover effect on them the next day.
- Sodium oxybate may have a role in worsening sleep breathing disorder in narcolepsy, particularly in obese patients or in patients with obstructive sleep apnea
- Sodium intake will be increased by 0.75 – 1.6 g/day in patients receiving sodium oxybate 4.5 – 9 g/day, and sodium intake reductions should be considered in patients with heart failure, hypertension, or impaired renal function.
Contraindications
Concurrent use of Xyrem (sodium oxybate) with sedative hypnotic agents or alcohol is contraindicated. Xyrem is contraindicated in patients with succinic semialdehyde dehydrogenase deficiency. This rare disorder is an inborn error of metabolism variably characterized by mental retardation, hypotonia, and ataxia, and causes GHB to accumulate in the blood.
References
1) Robinson DM, Keating GM. Sodium Oxybate – A review of its use in the management of narcolepsy. CNS Drugs 2007; 21(4): 337 – 354
2) Wang Y.G et al. Safety Overview of Postmarketing and Clinical Experience of Sodium Oxybate (Xyrem): Abuse, Misuse, Dependence, and Diversion. Journal of Clinical Sleep Medicine 2009; 5(4): 365 – 380
3) Nishino S. Okuro K. Emerging treatments for narcolepsy and its related disorders. Expert Opin. Emerging Drugs. (2010): 15(1)\
4) Canadian Pharmacists Association 2012. e-CPS Online.
5) http://www.xyrem.com/healthcare-professionals/direct-your-patients
Images:
Robinson DM, Keating GM. Sodium Oxybate – A review of its use in the management of narcolepsy. CNS Drugs 2007; 21(4): 337 – 354
http://bouldersleeptherapy.com/disorders/narcolepsy.php/
autoimmunediseasesa2z.com
www3.unil.ch
2) Wang Y.G et al. Safety Overview of Postmarketing and Clinical Experience of Sodium Oxybate (Xyrem): Abuse, Misuse, Dependence, and Diversion. Journal of Clinical Sleep Medicine 2009; 5(4): 365 – 380
3) Nishino S. Okuro K. Emerging treatments for narcolepsy and its related disorders. Expert Opin. Emerging Drugs. (2010): 15(1)\
4) Canadian Pharmacists Association 2012. e-CPS Online.
5) http://www.xyrem.com/healthcare-professionals/direct-your-patients
Images:
Robinson DM, Keating GM. Sodium Oxybate – A review of its use in the management of narcolepsy. CNS Drugs 2007; 21(4): 337 – 354
http://bouldersleeptherapy.com/disorders/narcolepsy.php/
autoimmunediseasesa2z.com
www3.unil.ch