Pathophysiology of narcolepsy
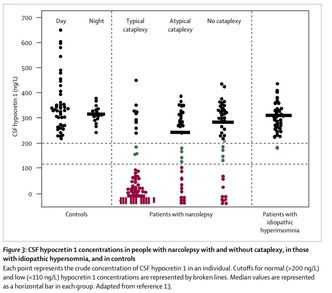
The deficiency of hypocretin-1
The pathophysiology of narcolepsy is thought to be due to the neurotransmitter hypocretin/orexin, and is believed to be responsible for the symptoms of narcolepsy. Current evidence suggests that most cases of narcolepsy with cataplexy are associated with a loss of hypocretin-containing neurons, and patients with narcolepsy usually have low or undetectable concentrations of hypocretin in cerebrospinal fluid (CSF). The hypocretin system is a major excitatory system that affects the activity of monoaminergic (i.e. dopamine, norepinephrine, serotonin, and histamine) and cholinergic systems, with major effects on vigilance or wakefulness states.
Hypocretin-containing neurons are found in the hypothalamus, and communicate with numerous brain nuclei to help with the regulation of sleep and alertness. Hypocretin-1 receptors are localized in the LDT/PPT, dorsal raphe nucleus, and locus coerulus (LC).
Hypocretin is a wakefulness-promoting neuropeptide that enhances activity in motor systems, and its secretion (i.e. its concentration in the CSF) reaches its peak during normal wakefulness and with movement or periods of increased psychomotor activity. It is thought to be involved in increasing muscle tone through activating the locus coerulus and raphe nuclei. During wakefulness, these neurons are responsible for increasing the activity of motor neurons through the release of the neurotransmitters norepinephrine and serotonin. Its firing activity then decreases significantly during non-REM and REM sleep.
Research: Hypocretin replacement therapy
Since one of the main pathophysiological factors underlying the development of narcolepsy is the loss of hypocretin and hypocretin-producing neurons, replacing these peptides would be the ideal therapy.
Supplementation with hypocretin-replacing therapies such as the direct use of hypocretin agonists and hypocretin neuron transplantation are currently under investigation in animal models.
Thus far, they have been effective when they were administered centrally in hypocretin-deficient mouse models. However, attempts at using hypocretin-based treatments after peripheral administration have been disappointing, since they do not cross the blood-brain barrier, where they need to reach in order to exert their effects.
Hypocretin supplementation and replacement therapies have also been conducted on narcoleptic canines. In one study by Fujiki et al, the investigators assessed the effects that IV administration of hypocretin-1 would have on cataplexy. Their results demonstrated that only a small portion of hypocretin-1 penetrates to the CNS after high doses of IV hypocretin-1. However, high-dose hypocretin-1 was found to produce only a short-acting anti-cataplectic effect
Therefore, development of hypocretin agonists that penetrate the blood-brain-barrier (i.e. synthetic small molecular agonists) will likely be necessary for use in humans.
The pathophysiology of narcolepsy is thought to be due to the neurotransmitter hypocretin/orexin, and is believed to be responsible for the symptoms of narcolepsy. Current evidence suggests that most cases of narcolepsy with cataplexy are associated with a loss of hypocretin-containing neurons, and patients with narcolepsy usually have low or undetectable concentrations of hypocretin in cerebrospinal fluid (CSF). The hypocretin system is a major excitatory system that affects the activity of monoaminergic (i.e. dopamine, norepinephrine, serotonin, and histamine) and cholinergic systems, with major effects on vigilance or wakefulness states.
Hypocretin-containing neurons are found in the hypothalamus, and communicate with numerous brain nuclei to help with the regulation of sleep and alertness. Hypocretin-1 receptors are localized in the LDT/PPT, dorsal raphe nucleus, and locus coerulus (LC).
Hypocretin is a wakefulness-promoting neuropeptide that enhances activity in motor systems, and its secretion (i.e. its concentration in the CSF) reaches its peak during normal wakefulness and with movement or periods of increased psychomotor activity. It is thought to be involved in increasing muscle tone through activating the locus coerulus and raphe nuclei. During wakefulness, these neurons are responsible for increasing the activity of motor neurons through the release of the neurotransmitters norepinephrine and serotonin. Its firing activity then decreases significantly during non-REM and REM sleep.
Research: Hypocretin replacement therapy
Since one of the main pathophysiological factors underlying the development of narcolepsy is the loss of hypocretin and hypocretin-producing neurons, replacing these peptides would be the ideal therapy.
Supplementation with hypocretin-replacing therapies such as the direct use of hypocretin agonists and hypocretin neuron transplantation are currently under investigation in animal models.
Thus far, they have been effective when they were administered centrally in hypocretin-deficient mouse models. However, attempts at using hypocretin-based treatments after peripheral administration have been disappointing, since they do not cross the blood-brain barrier, where they need to reach in order to exert their effects.
Hypocretin supplementation and replacement therapies have also been conducted on narcoleptic canines. In one study by Fujiki et al, the investigators assessed the effects that IV administration of hypocretin-1 would have on cataplexy. Their results demonstrated that only a small portion of hypocretin-1 penetrates to the CNS after high doses of IV hypocretin-1. However, high-dose hypocretin-1 was found to produce only a short-acting anti-cataplectic effect
Therefore, development of hypocretin agonists that penetrate the blood-brain-barrier (i.e. synthetic small molecular agonists) will likely be necessary for use in humans.
Neurobiology of cataplexy
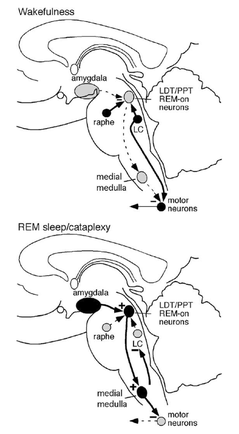
To understand the neurobiology of cataplexy, it is helpful to review the mechanisms that govern REM sleep (please also refer to the figure on the left):
During wakefulness:
Neurons that cause atonia (lack of normal muscle tone caused by a loss of motor neuron excitation) within the laterodorsal and pedunculopontine tegmental nuclei (LDT/PPT) are inhibited by the locus coerulus (LC) and raphe nuclei, which have noradrenergic and serotonergic activity. In addition, the LC directly increases the activity of motor neurons, thereby increasing muscle tone.
During REM sleep:
During REM sleep and probably during cataplexy, the LC and raphe nuclei are silent, allowing the LDT/PPT neurons in the dorsal pons to excite neurons of the medial medulla that directly inhibit motor neurons. The medial medulla also has an ascending projection that inhibits the LC neurons. The amygdala may be active during strong emotions, thus triggering an episode of cataplexy. Although descending excitatory pathways are still active during REM sleep, motor neuron inhibition during REM sleep is so profound that it suppresses all but the smallest movements
Therefore, the activation of the LDT/PPT drive the atonia of REM sleep and the manifestations of cataplexy.
Bottom line:
The role of emotions in triggering cataplexy:
An interesting but perplexing phenomenon is how cataplexy can be triggered by emotions such as laughter or anger. There are a few theories and postulations for explaining this observation:
During wakefulness:
Neurons that cause atonia (lack of normal muscle tone caused by a loss of motor neuron excitation) within the laterodorsal and pedunculopontine tegmental nuclei (LDT/PPT) are inhibited by the locus coerulus (LC) and raphe nuclei, which have noradrenergic and serotonergic activity. In addition, the LC directly increases the activity of motor neurons, thereby increasing muscle tone.
During REM sleep:
During REM sleep and probably during cataplexy, the LC and raphe nuclei are silent, allowing the LDT/PPT neurons in the dorsal pons to excite neurons of the medial medulla that directly inhibit motor neurons. The medial medulla also has an ascending projection that inhibits the LC neurons. The amygdala may be active during strong emotions, thus triggering an episode of cataplexy. Although descending excitatory pathways are still active during REM sleep, motor neuron inhibition during REM sleep is so profound that it suppresses all but the smallest movements
Therefore, the activation of the LDT/PPT drive the atonia of REM sleep and the manifestations of cataplexy.
Bottom line:
- Insufficient hypocretin levels results in an imbalance between motor excitation & inhibition triggered by emotions
- Inactivation of the excitatory system and activation of the inhibitory system
- Net result: decreased muscle tone leads to cataplexy
The role of emotions in triggering cataplexy:
An interesting but perplexing phenomenon is how cataplexy can be triggered by emotions such as laughter or anger. There are a few theories and postulations for explaining this observation:
- Neurons of the amygdala are often active during strong emotions, and stimulation of the amygdala can increase REM sleep, perhaps via its projections to the LDT/PPT and nearby regions
- In recordings from narcoleptic dogs, amygdala neurons often fire just before and during an episode of cataplexy, suggesting that they may help trigger the response.
- Activation of emotionally triggered events such as laughing may result in significant atonia lasting for several minutes in patients with narcolepsy with cataplexy
References
Content:
1. Houghton WC, Scammell TE, Thorpy M. Pharmacotherapy for cataplexy. Sleep Medicine Reviews 2004; 8: 355-3662.
2. Ahmed I, Thorpy M. Clinical features, diagnosis and treatment of narcolepsy. Clin Chest Med 2010; 31: 371 – 381
3. Watson CJ, et al. Sleep 2008;31:453-644) Nishino S. Clinical and neurobiological aspects of narcolepsy. Sleep Medicine 2007; 2: 373 – 399
5) Fujiki N, Ripley B, Yoshida Y, Mignot E, Nishino S. Effects of IV and ICV hypocretin-1 (orexin A) in hypocretin receptor-2 gene mutated narcoleptic dogs and IV hypocretin-1 replacement therapy in a hypocretin ligand deficient narcoleptic dog. Sleep 2003;6(8):953–9
Images:
Houghton WC, Scammell TE, Thorpy M. Pharmacotherapy for cataplexy. Sleep Medicine Reviews 2004; 8: 355-3662.
Dauvilliers Y., Arnulf I, Mignot E. Narcolepsy with Cataplexy. Lancet (Seminar) 2007; 369: 499 – 511
http://bouldersleeptherapy.com/disorders/narcolepsy.php/
autoimmunediseasesa2z.com
www3.unil.ch
